Dihydrotestosterone (DHT) is central to androgenetic alopecia, but it is far more than a “baldness hormone”. This article explains what DHT does in the body across a lifetime, how it affects hair follicles in men and women, the evidence linking DHT to pattern hair loss, and why reducing DHT is a rational strategy – within limits.
![[headshot]](https://cdn.prod.website-files.com/68207da82e5b8c350c67932f/68c6a7436a552c0fe87a4da7_Screenshot%202024-11-27%20at%2011.38.11.png)
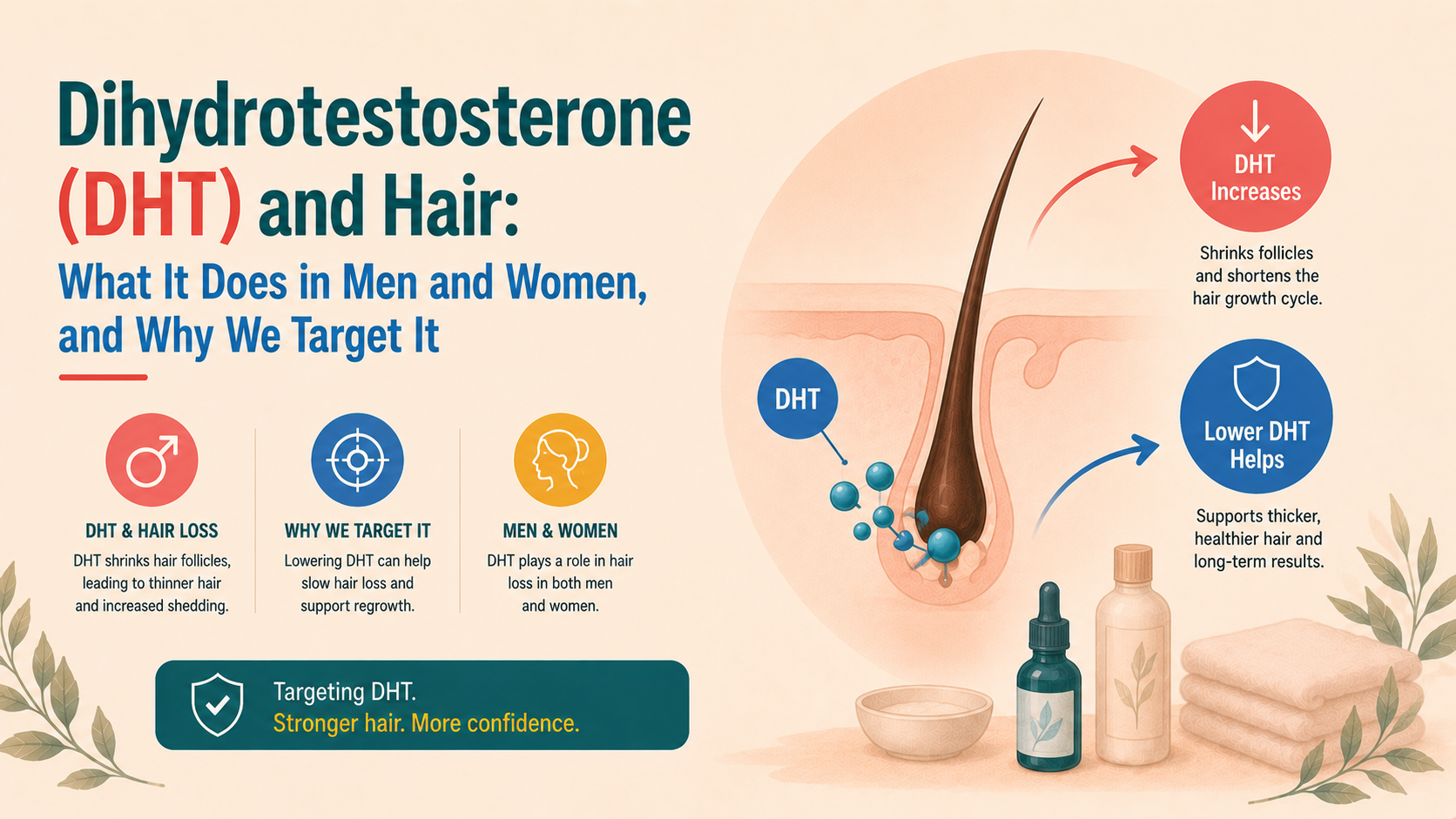
DHT is a normal androgen made from testosterone that plays an important role in early male development and puberty, but in adulthood it acts mainly as a local signal in tissues like the scalp, prostate and skin. In genetically susceptible individuals, DHT drives follicular miniaturisation on the scalp, shortening the growth phase and producing progressively thinner hairs, which explains the typical patterns of androgenetic alopecia. This effect depends less on how much DHT is in the bloodstream and more on how sensitive specific follicles are to it and how much local enzyme activity is present. Evidence from genetics, physiology and drug studies consistently shows that reducing DHT can slow hair loss and sometimes reverse miniaturisation, without removing core male functions in most men because testosterone continues to fulfil those roles. In women, the relationship is more nuanced, with DHT interacting alongside broader hormonal balance and follicular sensitivity. Overall, DHT is not inherently harmful, but in the wrong follicles and context it becomes a key driver of pattern hair loss, which is why targeting it forms a central part of treatment strategies.
When people begin to read about hair loss, they quickly run into three letters: DHT. It is blamed for receding hairlines, expanding crowns, oily skin and prostate problems. Online, it is either vilified as a toxin or defended as the essence of masculinity.
The reality is quieter and more interesting. DHT is:
Understanding DHT helps explain why some men and women lose hair the way they do, why 5α-reductase inhibitors can help, and where the limits and trade-offs lie.
Dihydrotestosterone is a steroid hormone from the androgen family. It is made when testosterone is converted by 5𝝰-reductase. There are at least two main isoforms of this enzyme relevant to hair and skin:
DHT binds to the androgen receptor more tightly than testosterone and activates it more strongly. In tissues where 5𝝰-reductase is active, DHT is the dominant androgenic signal.
Think of testosterone as the main circulating androgen, and DHT as a powerful local amplifier in specific organs where 5𝝰-reductase is active.
In the male fetus, DHT is essential for the normal development of the external genitalia. It helps the genital tubercle differentiate into a penis rather than a clitoris, shapes the scrotum and contributes to urethral closure.
Children born with a deficiency in the type II form of 5𝝰-reductase make very little DHT. Despite normal or near-normal testosterone, they may be born with ambiguous or feminised external genitalia. Puberty later reveals their underlying male biology as testosterone levels rise, but the early absence of DHT demonstrates its importance in genital formation.
At puberty, the testes produce more testosterone. In tissues with 5𝝰-reductase, DHT is generated in greater quantities and contributes to:
Testosterone and DHT together forge the external traits we recognise as male secondary sexual characteristics. If either is missing, the process is altered.
Once puberty is complete, the role of DHT changes.
Testosterone carries most of the weight for muscle mass, bone density, libido, and sperm production.
DHT continues to exert its influence mainly where 5𝝰-reductase is present in abundance: in the prostate, skin and hair follicles.
Studies of families with inherited 5𝝰-reductase deficiency, and of men treated with strong 5α-reductase inhibitors for years, show that:
After puberty has done its job, DHT is less about making a man a man and more about maintaining prostate size (sometimes a little too enthusiastically), driving acne and sebum production in predisposed individuals, and nudging vulnerable scalp follicles towards miniaturisation.
It is a powerful hormone in the wrong place, at the wrong time, in the wrong follicle.
Women also produce androgens from the ovaries, adrenals and via peripheral conversion in skin and fat. DHT in women has subtler roles but is not irrelevant.
In polycystic ovary syndrome (PCOS) and other hyperandrogenic states, total and free androgens are often elevated. Clinically, this can mean increased terminal hairs on the face, chest and abdomen, persistent acne, menstrual irregularity or anovulation and, for some, premature or more aggressive thinning through the mid-scalp.
However, many women with female pattern hair loss have entirely normal blood androgen levels. Here, the issue is less “too much DHT in the bloodstream” and more:
The effect of DHT on hair depends on where the follicle is located and how it is genetically wired.
In androgen-dependent sites like the beard, chest and pubic area, androgens (including DHT) stimulate vellus hairs to become thicker, pigmented terminal hairs. As a result, hair becomes coarser and growth more robust.
In androgen-sensitive sites on the scalp (frontal, temporal, and vertex regions in genetically predisposed people):
The back and sides of the scalp, in contrast, usually show:
This regional difference underpins both the pattern of baldness and the logic of transplanting hair from the occiput (back of the head) to the front.
Several lines of evidence point to DHT as a key driver of androgenetic alopecia.
The classic male pattern (receding hairline and vertex thinning with preserved occipital fringe) mirrors the distribution of androgen-sensitive vs resistant follicles.
Histological studies show increased 5𝝰-reductase expression and androgen receptor density in the balding frontal/vertex scalp compared with the occipital scalp in men with AGA.
Men with a lifelong deficiency of type II 5α-reductase rarely develop male pattern baldness. Their prostates are tiny, and their body hair is sparse, but their testosterone levels and gender identity are male.
Historically, castrated men (or men who lose their testes before puberty) do not develop typical male pattern baldness. Conversely, androgen administration or replacement in such men can induce androgenic hair changes.
Men taking finasteride or dutasteride – drugs that lower DHT by inhibiting 5α-reductase – show slower hair loss and, in many cases, partial regrowth compared with placebo.
Reductions in scalp DHT achieved with these drugs correlate with improvements in hair counts and reversal of miniaturisation in clinical trials.
Taken together (regional follicle biology, genetic deficiencies, castration data and drug trials), a coherent picture emerges: These observations do not mean DHT is the only cause, but they strongly implicate it.
In women, the relationship is more nuanced, but DHT still matters.
However, many women with FPHL have normal serum DHT and testosterone, and some women with biochemical hyperandrogenism never develop significant scalp thinning.
The difference lies in:
This is why some postmenopausal women, without obvious hyperandrogenism, notice accelerated thinning: oestrogen levels fall, the androgen:oestrogen ratio shifts, and the same DHT that was previously manageable now becomes more influential at the follicle.
Given all of this, reducing DHT (especially in the scalp) becomes a logical therapeutic target for androgenetic alopecia.
If DHT accelerates miniaturisation in susceptible follicles, then reducing DHT should slow that process and, where follicles are not irreversibly shrunken, allow some reversal.
This is exactly what is observed with:
Crucially, targeted DHT reduction does not remove testosterone, and therefore does not, in most men, strip away fundamental male characteristics. Blocking DHT is more like turning down the gain on a specific channel than muting the entire system.
Where DHT is doing more harm than good, for example, in prostatic overgrowth and androgenetic alopecia, that trade-off is often acceptable. Where DHT is still contributing usefully (foetal genital development, puberty), blocking it would be harmful. This is why 5𝝰-reductase inhibitors are contraindicated in women who may become pregnant, and are generally not suitable before puberty in men.
The widespread use of 5𝝰-reductase inhibitors (5-ARIs) for prostate enlargement and hair loss provides us with a long-term observation of what happens when human DHT is suppressed:
This empirical experience supports the idea that:
In women, we have less data, but enough to be cautious:
If you are a man with early or moderate pattern baldness, you now know that most of the mischief is occurring where DHT meets genetically sensitive follicles, that your occipital hair is naturally resistant, and that DHT is not the sole guardian of your manhood.
If you are a woman with FPHL or PCOS, then you know that DHT is a player, but the larger hormonal picture and follicular sensitivity matter just as much. Further, any systemic targeting of DHT has to be weighed against reproductive plans and broader endocrine health.
Understanding DHT raises the quality of the conversation from a simple explanation that a drug blocks a hormone that causes baldness to a more nuanced discussion of the biological changes taking place and the potential consequences involved.
Primary sources
Physiology and endocrine references
DHT and hair-specific studies
Women and DHT/FPHL
Therapeutic evidence as indirect support